- Blog
- Chromecast ps remote play
- Burning sage
- Remixvideo mac torrent
- Topaz gigapixel ai for video crack
- Resogun ps3
- Lumo orbital
- Zen meditation
- Table structure behind oracle workspace manager
- Where to buy red wing boots
- Boondocking in missouri
- Journey to the savage planet cheats
- Inddesign forum with incatalog
- Blog
- Chromecast ps remote play
- Burning sage
- Remixvideo mac torrent
- Topaz gigapixel ai for video crack
- Resogun ps3
- Lumo orbital
- Zen meditation
- Table structure behind oracle workspace manager
- Where to buy red wing boots
- Boondocking in missouri
- Journey to the savage planet cheats
- Inddesign forum with incatalog
#Lumo orbital how to
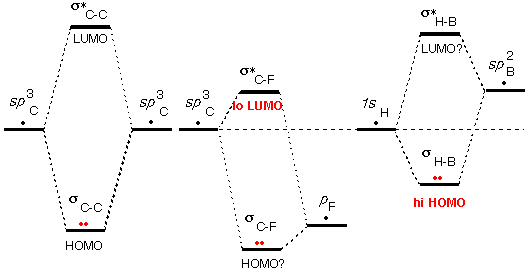
In this video I want to introduce a very important concept in organic chemistry called frontier molecular orbital theory, let's take a look, frontier orbital interactions are actually the riding force behind many reactions in organic chemistry, so this information that we're going to learn now it's actually going to be foundational to understand reactions later, so it's really important that we know what this FMOT theory is, okay? And the most important thing that we need to learn about frontier molecular orbitals is how to find HOMO and LUMO, so you might have heard about HOMO and LUMO before but in this lesson we really need to fully understand it, so the HOMO is defined as the highest occupied molecular orbital, so out of all your molecular orbitals it's the one that has the highest energy that still has electrons in it, okay? then there's the LUMO, the LUMO is the lowest unoccupied molecular orbital, so once again, out of all you molecular orbitals, it's the one that has the lower energy while still having no electrons in it, okay? So, if we were to look at ethene as an example, so ethene is a very simple molecule so it's easy to understand HOMO, LUMO. Orbital Diagram: 6-atoms- 1,3,5-hexatriene This video shows you how to draw the different molecular orbitals for a conjugated system, how to make sense of the logic without the crazy math, and how to quickly and easily identify HOMO and LUMO molecular orbitals.Conjugated Hydrohalogenation (1,2 vs 1,4 addition) The electron can go back to it’s original orbital and heat (or light) is produced in the process. Video 2: HOMO and LUMO for Conjugated Systems An electron can be excited from the HOMO to the LUMO using light of a precise wavelength dependent on the energy difference between the two orbitals (since the orbitals are quantized). This video provides you with a logic-based overview of MO theory with simple take-aways to focus on at the organic chemistry level. Video 1: Molecular Orbital Theory for Sigma and Pi Bonds MO theory typically comes up when studying reactions of conjugated systems, especially when asked to identify HOMO and LUMO molecular orbitals for molecules in Diels-Alder and other pericyclic reactions. the Highest Occupied, HOMO and Lowest Unoccupied, LUMO). Molecular Orbital theory is a topic that comes up in general chemistry in relation to bond formation and bonding energy.Īt the organic chemistry level, we’re less concerned with the nitty-gritty quantum physics and crazy math and more interested in making just enough sense of the information to be able to apply what we know to reactions and mechanisms. Woodward and Hoffmann showed that by examining the interaction of the frontier molecular orbitals (ie.
